![]()
CONQUER is contributing invaluable data to the search for a cure for scleroderma.
CONQUER, a Groundbreaking Scleroderma Registry
In this video, learn why scleroderma patient participation in the registry is essential for improving care and developing more effective, personalized therapies for scleroderma patients.
In 2018, the SRF launched the CONQUER Registry (an acronym for COllaborative National QUality and Efficacy Registry) – a first-of-its-kind nationwide scleroderma patient registry and biosample repository aimed at improving care and developing more effective, personalized therapies for systemic sclerosis (scleroderma) patients.
CONQUER was created through a collaborative effort among the largest scleroderma research centers in the U.S. With the goal of enrolling thousands of scleroderma patients from around the country, researchers and clinicians will be able to learn as much as possible about all forms of scleroderma in patients of all backgrounds and ethnicities and use that knowledge to advance scleroderma research and discovery.
As of December 2025, more than 1,300 patients have enrolled in CONQUER, contributing invaluable data to the search for a cure for scleroderma.
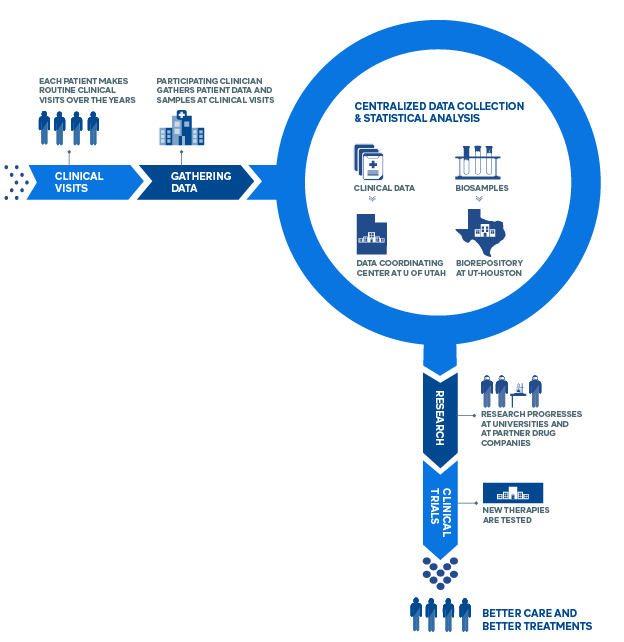
How the CONQUER Registry Works
- Scleroderma patients participate through their routine clinical visits (2x/year), contributing Clinical data & Blood samples
- Scleroderma patients also contribute data through Patient-Reported Outcome surveys. (This can be done at the patient’s convenience.)
- Enrolled scleroderma patients receive the most modern standard of care. There are no experimental therapies involved in CONQUER.
Who is Eligible to Participate?
To be eligible for CONQUER, the national scleroderma patient registry, you must:
- Be 18 years of age or older
- Have systemic scleroderma
- Be within first 5 years of first non-Raynaud’s symptom (Enrollment is time sensitive)
You can participate in CONQUER even if you are enrolled in another registry.
CONQUER will benefit every scleroderma patient, even those unable to enroll.
How Do I Participate in CONQUER?
- The 19 institutions listed below are the sites participating in CONQUER.
- We do anticipate that other expert sites will be added over time and are in the process of evaluating such additions now. If you are a patient at one of these participating centers, ask your doctor about enrolling in CONQUER.
- Otherwise, please contact the Scleroderma Research Foundation at info@srfcure.org or call us at 800.441.CURE (2873).
CONQUER Sites
Boston University Medical Center
Principal Investigator: Andrea Bujor, MD, PhD
Research Coordinators:
Ben Chamis
bchamis@bu.edu
Zeinab Diallo
zeinabd@bu.edu
Columbia University, New York
Principal Investigator: Elana Bernstein, MD, MSc
Co-Investigator: Yiming Luo, MD
Research Coordinators:
Niki Pradhan
np2749@cumc.columbia.edu
Claudia Salazar
cs4559@cumc.columbia.edu
Edna Diarte Acosta
eyd2114@cumc.columbia.edu
Roger Lent
rl3646@cumc.columbia.edu
212.342.6815
Georgetown University, Washington, D. C.
Principal Investigator: Virginia Steen, MD
Research Coordinator: Maia Cecire
mz381@georgetown.edu
Hospital for Special Surgery, New York
Principal Investigator: Jessica Gordon, MD
Co-Investigators: Kim Lakin, MD, MS; Robert Spiera, MD
Research Coordinators:
Oshmita Golam
golamo@hss.edu
Liza Morales
moralesli@hss.edu
212.774.7620
Duke University, North Carolina
Principal Investigator: Ankoor Shah, MD
Research Coordinators:
Karissa Grier
karissa.grier@duke.edu
Edna Scarlett
edna.scarlett@duke.edu
Johns Hopkins University, Maryland
Principal Investigators: Ami Shah, MD, and Laura Hummers, MD
Research Coordinators:
Adrianne Woods
awoods9@jhmi.edu
Cheryl Clare
cdaniel9@jh.edu
Gwen Leatherman
gleathe@jhmi.edu
Michelle Leatherman
mleathe4@jhmi.edu
410.550.6820
Mass General Hospital, Massachusetts
Principal Investigator: Flavia Castelino, MD
Research Coordinators:
Charles Toulmin
ctoulmin@mgh.harvard.edu
Ana Fernandes
adfernandes@mgh.harvard.edu
507.923.0814
Mayo Clinic – Rochester, Minnesota
Principal Investigator: Ashima Makol, MD
Research Coordinator: Jennifer Sletten, Research Coordinator
Sletten.Jennifer@mayo.edu
507.284.4797
Medical University of South Carolina
Principal Investigator: Faye Hant, DO, MSCR
Research Coordinator: Miranda Irvin
irvinm@musc.edu
Northwestern University, Illinois
Principal Investigator: Carrie L. Richardson, MD
Co-Investigator: Duncan Moore, MD
Research Coordinators:
Alexandra Soriano
alexandra.soriano@northwestern.edu
Kathleen Aren
kathleen.aren@northwestern.edu
312.503.1120
Stanford University, California
Principal Investigator: Lori Chung, MD, MS
Research Coordinators:
Kathryn Jee
kjee@stanford.edu
Swarna Nandyala
swarnan@stanford.edu
The University of Texas Health Science Center at Houston
Principal Investigator: Shervin Assassi, MD
Research Coordinators:
Maya Washington
Maya.Washington@uth.tmc.edu
Samuel “Sam” Theodore
samuel.theodore@uth.tmc.edu
University of California, Los Angeles
Principal Investigator: Elizabeth Volkmann, MD, MS
EVolkmann@mednet.ucla.edu
Research Coordinator: Nahal Ghanoongooi
nghanoongooi@mednet.ucla.edu
University of Colorado, Anshutz Medical
Principal Investigator: Melisssa Griffith, MD
melissa.griffith@cuanschutz.edu
Research Coordinators:
Kristin Sturm
kristin.sturm@cuanschutz.edu
Samantha Cotto
samantha.cotto@cuanschutz.edu
Vanessa Hernandez
vanessa.2.hernandez@cuanschutz.edu
Katerina Popke
katerina.popke@cuanschutz.edu
University of Michigan
Principal Investigator: Dinesh Khanna, MD
Research Coordinator:
Amisha Kaur
amikaur@med.umich.edu
University of Minnesota
Principal Investigator: Jerry Molitor, MD
Research Coordinators:
Katherine Wu
wuxx1320@umn.edu
Zoe Wright
wrig1316@umn.edu
612.624.5617
University of Pennsylvania
Principal Investigator: Nora Sandorfi, MD
Research Coordinators:
Amanda Shini
Amanda.Shini@Pennmedicine.upenn.edu
Lauren Baker
Lauren.Baker@Pennmedicine.upenn.edu
215.614.4430
University of Utah
Principal Investigator: Dorota Lebiedz-Odrobina, MD
Research Coordinators:
Inge Stijleman
inge.stijleman@hsc.utah.edu
Sally Bradstreet
Sally.Bradstreet@hsc.utah.edu
801.213.1368
Vanderbilt University Medical Center, Tennessee
Principal Investigator: Tracy Frech, MD
Research Coordinators:
Sarah Wood
sarah.wood@vumc.org
Utkir Bek
u.abdumominov@vumc.org
615.322.3635
Why Tatiana Participates in CONQUER
“Being included in this study gives me comfort and a sense of purpose, knowing that my experiences may help others one day.”
Read more about her journey with scleroderma and why research is important to her.

How Does My Data Get Used, and Is It Protected?
- Your clinical data and blood samples will be de-identified so no one can get personal information about you.
- CONQUER data is protected in a secure data center at the University of Utah. This Data Coordinating Center (DCC), based at the University of Utah School of Medicine, provides a central repository for data generated by each of the CONQUER sites. The DCC also works with each of the CONQUER Principal Investigators and Research Coordinators to implement CONQUER-wide standards for data collection and analysis in order to ensure uniformity and quality of the data. The DCC implements regular monitoring activities and performs statistical analyses for many manuscripts published on behalf of CONQUER.
- CONQUER blood samples are stored separately at the University of Texas-Houston, the largest biorepository of scleroderma samples in the world.
What is the Benefit to Me?
- Your participation will help accelerate advances in clinical care
- This registry supports the development of new therapies
- Patients enrolled in CONQUER will experience heightened scrutiny and care in a formal setting
- In the future, CONQUER will include a clinical trials platform for rapid testing of new drugs and enrolled patients may be eligible to participate in these trials.
WEBINAR SERIES
Learn About CONQUER
Watch an informative webinar explaining the importance of the CONQUER Registry and how it could lead to finding a cure for scleroderma.

What Have We Learned From CONQUER So Far?
Researchers have already published papers based on analysis of CONQUER data.
Collaborative National Quality and Efficacy Registry (CONQUER) for Scleroderma: Outcomes from a Multicenter US-based Systemic Sclerosis Registry (2020)
By: Victoria K. Shanmugam, Tracy M. Frech, Virginia D. Steen, Laura K. Hummers, Ami A. Shah, Elana J. Bernstein, Dinesh Khanna, Jessica K. Gordon, Flavia V. Castelino, Lorinda Chung, Faye N. Hant, Emily Startup, John M. VanBuren, Luke B. Evnin & Shervin Assassi
Does hand involvement in Systemic Sclerosis limit PRO completion? (2021)
By: Tracy M. Frech, John M. VanBuren, Emily Startup, Shervin Assassi, Elana J. Bernstein, Flavia V. Castelino, Lorinda Chung, Chase Correia, Jessica K. Gordon, Faye N. Hant, Laura Hummers, Dinesh Khanna, Nora Sandorfi, Ami A. Shah, Victoria K. Shanmugam, Virginia Steen & Luke Evnin
Summary: The objective of this analysis is to examine whether the severity of systemic sclerosis (SSc)-hand involvement influences patient-reported outcome measure (PROM) completion rate. There were 339 CONQUER participants with total of 600 baseline and follow-up visits available for analysis. Fingertip resorption (acro-osteolysis) and calcium deposits (calcinosis) were associated with lower PROM completion, independent of the length of the questionnaires or the modality of administration (electronic or paper). Multiple strategies are needed to ensure optimal completion of PROM in longitudinal cohort studies.
Baseline Characteristics of Systemic Sclerosis Patients with Restrictive Lung Disease in a Multi-Center US Based Longitudinal Registry (2022)
By: Flavia V. Castelino, John M. VanBuren, Emily Startup, Shervin Assassi, Elana J. Bernstein, Lorinda Chung, Chase Correia, Luke B. Evnin, Tracy M. Frech, Jessica K. Gordon, Faye N. Hant, Laura K. Hummers, Dinesh Khanna, Nora Sandorfi, Ami A. Shah, Victoria K. Shanmugam, Virginia Steen
Summary: The researchers looked at the various characteristics associated with restrictive lung disease (RLD) in patients enrolled in CONQUER. There were no differences in age, gender or disease duration. Patients with restrictive lung disease had a mean disease duration of 2.6 years. Non-white race, higher physician global health assessment scores and dyspnea scores were independently associated with RLD. Interestingly, 45% of CONQUER participants already had RLD, highlighting the importance of screening for lung disease at initial diagnosis of systemic sclerosis.
Computed Tomography of the Chest to Screen for Interstitial Lung Disease in Patients with Systemic Sclerosis at Expert Scleroderma Centers in the U.S. (2022)
By: Elana J. Bernstein, Shervin Assassi, Flavia V. Castelino, Lorinda Chung, Chase Correia, Luke B. Evnin, Tracy M. Frech, Jessica K. Gordon, Brian A. Skaug, Faye N. Hant, Laura K. Hummers, Nora Sandorfi, Ami A. Shah, Victoria K. Shanmugam, Virginia D. Steen, Dinesh Khanna
Summary: Although a chest CT scan is the gold standard test for the diagnosis of interstitial lung disease, there is no consensus among rheumatologists regarding the use of chest CT to screen for interstitial lung disease in their patients with systemic sclerosis. The goal of this study was to describe the chest CT ordering practices at expert scleroderma centers in the United States and to determine which patient characteristics are associated with obtaining a chest CT. The researchers found that 80% of systemic sclerosis patients enrolled in CONQUER underwent a chest CT. Patients with a positive centromere antibody were less likely to have undergone a chest CT.
CONQUER Scleroderma: Association of Gastrointestinal Tract Symptoms in Early Disease with Resource Utilization (2023)
By: Sarah Luebker, Tracy M Frech, Shervin Assassi, Brian Skaug, Jessica K Gordon, Kimberly Lakin, Elana J Bernstein, Yiming Luo, Virginia D Steen, Ami A Shah, Laura K Hummers, Carrie Richardson, Duncan F Moore, Dinesh Khanna, Flavia V Castelino, Lorinda Chung, Puneet Kapoor, Faye N Hant, Victoria K Shanmugam, John M VanBuren, Jessica Alvey, Monica Harding, Ankoor Shah, Ashima Makol, Dorota Lebiedz-Odrobina, Julie K Thomas, Elizabeth R Volkmann, Jerry A Molitor, Nora Sandorfi
Summary: This report from the CONQUER cohort looked at how gastrointestinal tract (GIT) symptoms impacts health-related costs of care. Among the 211 CONQUER participants in this study, most (64%) had mild GIT symptoms, 26% had moderate symptoms, and 10% severe GIT symptoms at 12-months of follow-up. More upper endoscopy procedures and inpatient hospitalization occurred in the CONQUER participants with severe GIT symptoms. It is especially important to understand symptom activity prior to damage to the GIT and how diagnosis increases care costs.
The Collaborative National Quality and Efficacy Registry for Scleroderma: Association of Medication Use on Gastrointestinal Tract Symptoms in Early Disease and the Importance of Tobacco Cessation (2023)
By: S. Luebker, T.M. Frech, S. Assassi, J.K. Gordon, E.J. Bernstein, V.D. Steen,
A.A. Shah, L.K. Hummers, C. Richardson, D. Khanna, F.V. Castelino1, L. Chung, F.N. Hant, V.K. Shanmugam, J.M. VanBuren, J. Alvey, M. Harding, N. Sandorfi
Summary: This manuscript presents the gastrointestinal tract (GIT) outcomes in relation to other scleroderma manifestations and medication exposures. 415 enrolled CONQUER participants were included in this analysis. Most participants had stable mild GIT symptoms at baseline and were prescribed immunosuppression and anti-heartburn therapy. In most patients, anti-reflux medication and immunosuppression initiation preceded the baseline visit, whereas anti-fibrotic initiation occurred at or after the baseline visit. There was no clear association between immunosuppression or anti-fibrotic use and severity of GIT symptoms. However, active tobacco use was associated with worse GIT symptoms, highlighting the importance of smoking cessation counselling.
Psychometric Evaluation of the Scleroderma Skin Questionnaire: A Novel Patient-Reported Outcome for Skin Disease in Patients With Systemic Sclerosis (2025)
By: Jeong Min Yu, John M VanBuren, Angela Child, Jessica S Alvey, Lisa A Mandl, Laura C Pinheiro, Shervin Assassi, Elana J Bernstein, Flavia V Castelino, Lorinda Chung, Luke Evnin, Tracy M Frech, Faye N Hant, Laura K Hummers, Dinesh Khanna, Kimberly S Lakin, Dorota Lebiedz-Odrobina, Yiming Luo, Ashima Makol, Jerry A Molitor, Duncan F Moore, Carrie Richardson, Nora Sandorfi, Ami A Shah, Ankoor Shah, Victoria K Shanmugam, Brian Skaug, Virginia D Steen, Elizabeth R Volkmann, Jessica K Gordon
Use of Nintedanib for the Treatment of Systemic Sclerosis-associated Interstitial Lung Disease at Expert Scleroderma Centers in the United States (2025)
By: Yiming Luo, Shervin Assassi, Maureen D Mayes, Tracy M Frech, Virginia D Steen, Carrie Richardson, Lorinda Chung, Nora Sandorfi, John M VanBuren, Monica Harding, Jessica S Alvey, Brian Skaug, Zsuzsanna McMahan, Kimberly S Lakin, Carleigh Zahn, Sonali J Bracken, Dorota J Lebiedz-Odrobina, Jerry A Molitor, Luke B Evnin, Duncan F Moore, Ankoor Shah, Flavia V Castelino, Elizabeth R Volkmann, Faye N Hant, Ashima Makol, Ami A Shah, Laura K Hummers, Jessica K Gordon, Dinesh Khanna, Elana J Bernstein
Hand swelling and other non-Raynaud's symptoms as the initial presentation of systemic sclerosis: prevalence and clinical associations in two U.S. cohorts (2025)
By: Iqtidar Hanif, Shervin Assassi, Maureen D. Mayes, Zsuzsanna H. McMahan, Meng Zhang, Julio Charles, John M. VanBuren, Jessica S. Alvey, Kimia Ghaffari, Elana J. Bernstein, Flavia V. Castelino, Lorinda Chung, Luke Evnin, Tracy M. Frech, Jessica K. Gordon, Faye N. Hant, Laura K. Hummers, Dinesh Khanna, Kimberly S. Lakin, Dorota Lebiedz-Odrobina, Yiming Luo, Ashima Makol, Jerry A. Molitor, Duncan F. Moore, Carrie Richardson, Nora Sandorfi, Ami A. Shah, Ankoor Shah, Victoria K. Shanmugam, Virginia D. Steen, Elizabeth R. Volkmann, Carleigh Zahn, Brian Skaug
Summary: This study indicates that for over 30% of people with systemic sclerosis, the earliest symptoms weren’t Raynaud’s phenomenon; instead, signs like puffy fingers or hand swelling came first.
Still Want More Information About Conquer?
- Download our flier here
- Email info@srfcure.org for any questions you may have.
OUR RESEARCH
Dive Deeper into Our Research
Dig into the details of our scleroderma research program to understand more about the science behind what our elite researchers are doing to find better treatments and, ultimately, a cure.
CONQUER Registry Sponsors







